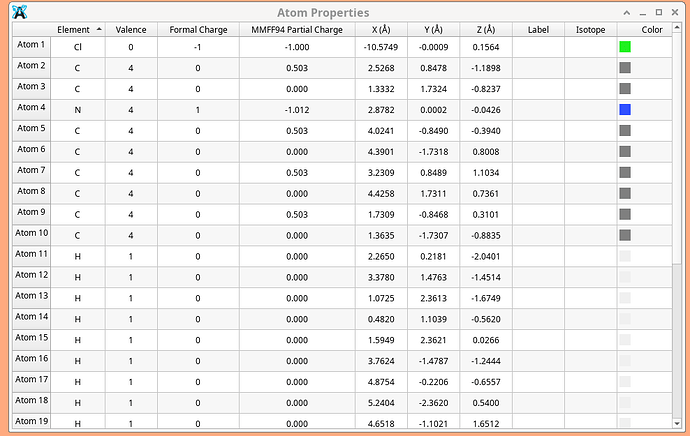
I am looking at ionic materials and finding strange results. I downloaded the structure for tetraethylammonium chloride. I then asked for an analysis. The table below has the nitrogen more negative (partial charge, not formal) than the chloride, with 4 carbons bearing a half positive charge each. That doesn’t seem reasonable to me given that the whole tetraethyl ammonium ion should just have a total charge of +1. FWTIW
I’m not sure I’d put a lot into considering the partial charges from MMFF94. I’d probably suggest something like GFN2-XTB if you want something fast but more accurate.
But “buried” nitrogens are notoriously challenging to interpret as far as partial charges. Consider that whatever electron density around the N and the carbons has to get divided up across 4 carbon atoms and the nitrogen. How you do that will determine what charge goes on what atom.
Thank you for your reply. I don’t know enough about these force fields yet to choose one. I took the default. I will go back and try again. Thanks,
John
Well, N is an electronegative element…
I have seen this before with other charge definitions, where for example in NH4+ (by itself) the positive charge is exclusively on the hydrogen atoms.
True,but look at the separation of charge. The carbons total 2+ charges, while the N has more (-) charge than the chloride. Nitrogen’s electronegativity is 0.7 vs Cl at 3.16 and the N has more negative charge? Seems mighty difficult to explain.